Building a Defensible Clinical Literature Review
Do’s, Don’ts, and Best Practices
“The distinction between a defensible clinical evaluation and a regulatory setback often lies in the quality of the literature review process.”
by Dr
Nadhim
Bayatti
Senior Analyst, SciMed Consultancy Ltd
EXECUTIVE SUMMARY
A robust clinical literature review is essential for compliance with EU MDR and IVDR requirements.
It must be structured, reproducible, and scientifically rigorous - not just a collection of publications.
Using frameworks such as PICO and predefined protocols ensures clarity, reduces bias, and improves consistency.
Systematic screening and critical appraisal are necessary to generate credible and defensible clinical evidence.
Poorly executed literature reviews are a common cause of regulatory delays and compliance issues.
Following clear “do’s and don’ts” helps avoid common pitfalls and strengthens submissions.
When done correctly, a literature review becomes a strategic regulatory asset rather than a liability.
INTRODUCTION
Under the European Union Medical Device Regulation (EU MDR) and In Vitro Diagnostic Regulation (IVDR), manufacturers are required to demonstrate robust clinical evidence supporting safety, performance, and clinical benefit. Clinical evaluation is a continuous, lifecycle-driven process that demands scientific rigour, traceability, and transparency.
A well-executed clinical literature review sits at the centre of this requirement. It must be:
Structured - based on a predefined methodology and protocol
Reproducible - another reviewer should reach comparable results using the same process
Aligned with regulatory guidance, including Medical Device Coordination Group (MDCG) guidance (notably MDCG 2020-6) and International Medical Device Regulators Forum (IMDRF) frameworks
The objective is not simply to ‘find papers’, but to systematically identify, appraise, and analyse clinical data relevant to the device under evaluation.
INDUSTRY CONTEXT & BACKGROUND
A poorly planned literature review often results in:
Insufficient or irrelevant clinical evidence
Over-reliance on low-quality publications
Inability to demonstrate state of the art
Delays during notified body review
Requests for additional clinical investigations
Many compliance challenges arise not from a lack of data, but from poorly designed literature strategies. The distinction between a defensible clinical evaluation and a regulatory setback often lies in the quality of the literature review process.
WHAT IS A LITERATURE SEARCH?
A clinical literature search is the systematic identification, selection, and critical appraisal of published clinical evidence relevant to a medical device. It supports:
Demonstration of safety and performance
Confirmation of clinical benefit
Establishment of state of the art
Identification of residual risks
Inputs into post-market surveillance
Unlike a narrative review, it follows a structured approach closer to a systematic review, even if it does not fully meet academic systematic review standards.
CORE COMPONENTS
Identification of Clinical Evidence
Relevant studies on:
The device under evaluation
Equivalent devices (if applicable)
Similar technologies
Alternative treatments (State of the Art)
Systematic Screening
Based on predefined inclusion and exclusion criteria
Critical Appraisal
Assessment of methodological quality, bias, and relevance
THE PICO MODEL
An effective literature search strategy should be based on the PICO framework:
P - Population (e.g. adults with atrial fibrillation)
I - Intervention (the device or technology)
C - Comparator (standard treatment or alternative device)
O - Outcomes (clinical endpoints such as safety, accuracy, performance)
Using PICO:
Clarifies scope
Reduces bias
Aligns with intended purpose
Improves reproducibility
Without this structure, searches often become too broad (yielding irrelevant results) or too narrow (missing key evidence).
Get Practical MDR & IVDR Evidence Guidance
MedTech Horizon breaks down literature review methodology, clinical evidence strategy, and regulatory interpretation, based on what Notified Bodies are actually challenging today.
If you're responsible for Clinical Evaluation, this is designed for your day-to-day work.
PRACTICALITIES: HOW TO DO IT PROPERLY
Do: Use Robust Databases
Use established scientific databases with broad coverage of peer-reviewed clinical literature, such as:
PubMed
PubMed Central (PMC)
EMBASE
It is important to note that the Cochrane Collaboration database is indexed within PubMed, meaning systematic reviews and meta-analyses can be captured there. For device-under-evaluation searches, PMC can be particularly valuable because it provides access to full-text articles.
At a minimum, 2 types of searches must be carried out:
One for the device under evaluation
One state-of-the-art search on the generic device group and similar devices
Do: Predefine and Document Your Search Strategy
Your protocol should clearly define:
Databases used
Search strings and Boolean operators
Date ranges
Inclusion/exclusion criteria
Screening methodology
This ensures reproducibility, a key regulatory expectation.
Do: Screen in Phases
A structured screening process typically includes:
Title screening - removing clearly irrelevant records
Abstract review - assessing preliminary relevance
Methodological and applicability assessment - with justified exclusions
Full-text review - enabling deeper analysis of:
Study methodology
Statistical analysis
Adverse events
Device-specific details
Relying solely on abstracts presents significant risk.
Do: Appraise for Quality and Relevance
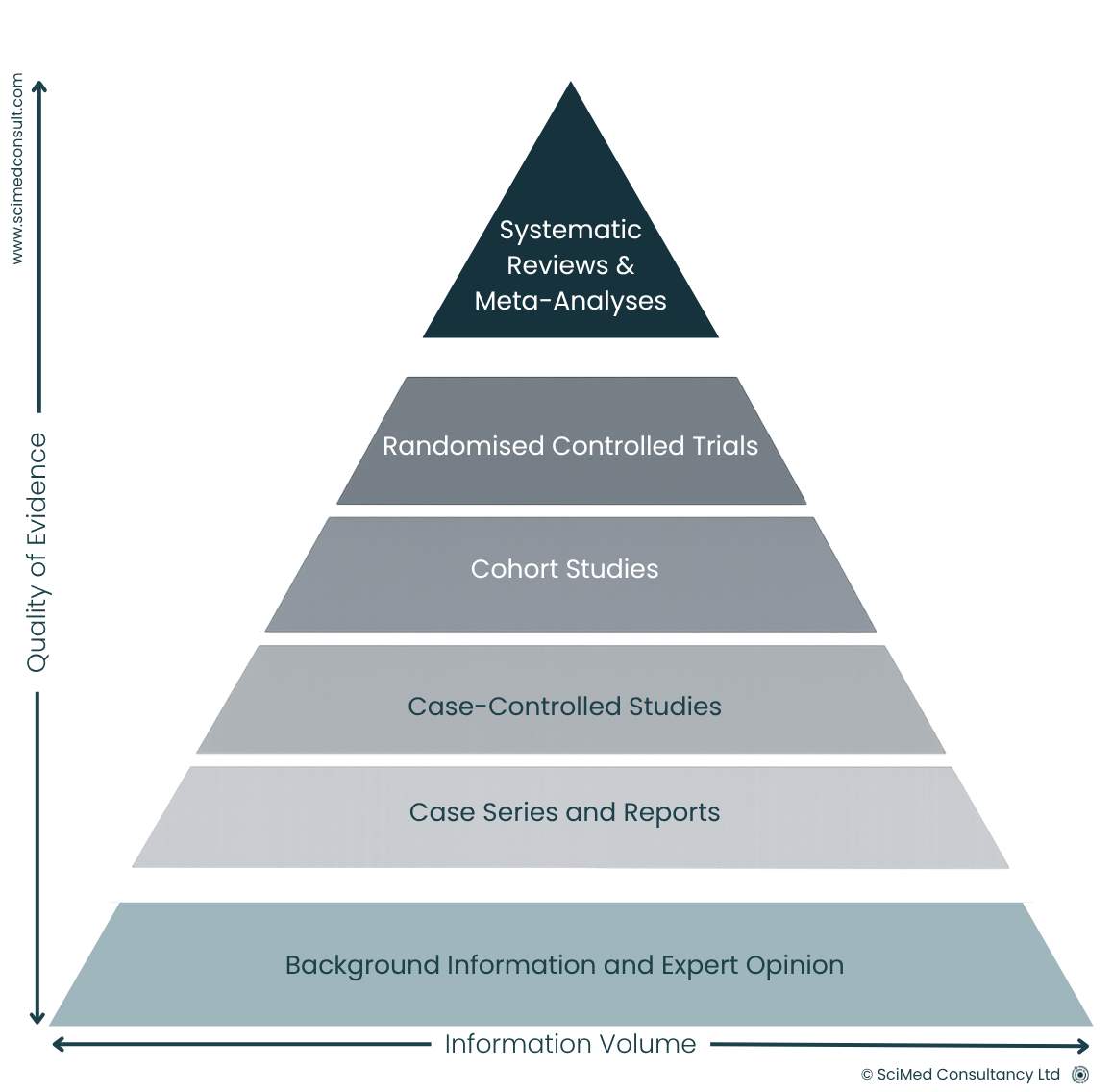
Not all evidence carries equal weight. Use clinical evidence ranking as described in MDCG 2020-6.
Higher levels of evidence:
Randomised controlled trials
Systematic reviews and meta-analyses
Lower levels of evidence:
Case series
Expert opinions
However, relevance is equally critical. A high-quality study on a different device may be less useful than a well-designed study on the actual device under evaluation. Key considerations:
Alignment of study population with intended use
Device equivalence
Clinical relevance of endpoints
Management of bias and confounding
DON’TS: COMMON PITFALLS
Don’t: Start Without a Plan
Unstructured searches lead to:
Missed publications
Inconsistent decisions
Regulatory findings during audits
Always define the protocol before starting.
Don’t: Rely Solely on Manufacturer Data
Over-reliance on company-sponsored studies and marketing-driven publications can undermine credibility. Balanced, transparent evidence is essential.
Don’t: Ignore Negative Evidence
Regulators expect:
Identification of adverse outcomes
Discussion of complications
Balanced benefit-risk assessment
Excluding contradictory evidence can invalidate the evaluation.
Don’t: Confuse Quantity with Quality
A large number of weak studies does not equal strong evidence. A few case reports cannot replace one well-conducted randomized trial.
Don’t: Treat Literature Review as a One-Time Activity
Clinical evaluation is continuous under EU MDR and IVDR. Literature review must feed into:
Post-Market Clinical Follow-up (PMCF)
Periodic Safety Update Reports (PSUR)
Risk management updates
PRESSURE-TEST YOUR LITERATURE REVIEW BEFORE SUBMISSION
Even well-structured literature reviews can fail under audit if methodology, screening logic, or evidence weighting is not clearly defensible.
Use our MDR Clinical Evaluation Audit-Readiness Checklist to assess whether your literature review process would stand up to Notified Body scrutiny, before it’s reviewed externally.
CONCLUSION
A clinical literature review for medical devices is both a scientific and regulatory exercise, not merely an academic formality.
To meet EU MDR and IVDR requirements, it must be:
Structured
Systematic
Reproducible
Critically appraised
Aligned with MDCG and IMDRF guidance
The Essential Do’s
✔ Base your search on a PICO framework
✔ Use recognised databases such as PubMed and PMC
✔ Document search strategy and screening decisions
✔ Critically appraise both quality and relevance
✔ Apply evidence hierarchy from MDCG 2020-6
✔ Include both positive and negative findings
✔ Maintain the review throughout the product lifecycle
The Critical Don’ts
✘ Don’t conduct ad hoc or undocumented searches
✘ Don’t rely only on abstracts
✘ Don’t overstate weak evidence
✘ Don’t ignore conflicting data
✘ Don’t treat literature review as a one-time submission task
When executed correctly, a literature review becomes a powerful regulatory asset. When done poorly, it becomes a liability. In today’s stricter regulatory landscape, robust clinical evidence is not optional.
When executed correctly, a literature review becomes a powerful regulatory asset. When done poorly, it becomes a liability. In today’s stricter regulatory landscape, robust clinical evidence is not optional.
USEFUL REFERENCES
MDCG 2020-6
NOT SURE YOUR LITERATURE REVIEW WOULD HOLD UP AT AUDIT?
Many literature reviews appear methodologically sound but fail when reviewers test reproducibility, relevance, and weighting logic.
A focused review can quickly show:
Whether your search strategy is reproducible
If your inclusion/exclusion logic is defensible
Where weak or misaligned evidence may undermine conclusions