Lifecycle Compliance: Maintaining MDR & IVDR-Ready Documentation
How LifeScan maintain compliant documentation through structured post-market support.
Project Type: Ongoing Literature Monitoring and SotA Updates
INTRODUCTION
Getting Clinical Evaluation and Performance Evaluation documentation across the line for MDR and IVDR is a significant achievement. Most teams breathe a quiet sigh of relief when it is done.
Then the question hits: how do we keep it there?
For LifeScan, a global manufacturer of glucose monitoring technology, that question surfaced almost immediately after the initial implementation work was complete.
SciMed had supported the organisation through the initial development of its evaluation documentation framework, so the focus now shifted to maintaining it.
The documentation framework was solid. The evidence base was defensible. But regulations continue to evolve, products mature, and notified bodies and auditors do not stop asking questions.
Maintaining compliant Clinical and Performance Evaluation documentation over the full product lifecycle is a different challenge entirely from building it in the first place, and LifeScan needed a structured answer.
For more guidance on Clinical and Performance Evaluation under MDR and IVDR, explore our Regulatory Insights.
KEY TAKEAWAYS: How SciMed Maintained LifeScan’s MDR & IVDR Compliance
· Established a structured lifecycle maintenance programme for Performance and Clinical Evaluation documentation.
· Introduced quarterly monitoring alongside annual updates to ensure documentation always remains regulator-ready, and literature searches no more than 3 months old.
· Audit responses are faster and more consistent due to a mature documentation process.
· The SciMed team operates as an extension of LifeScan’s clinical operations function.
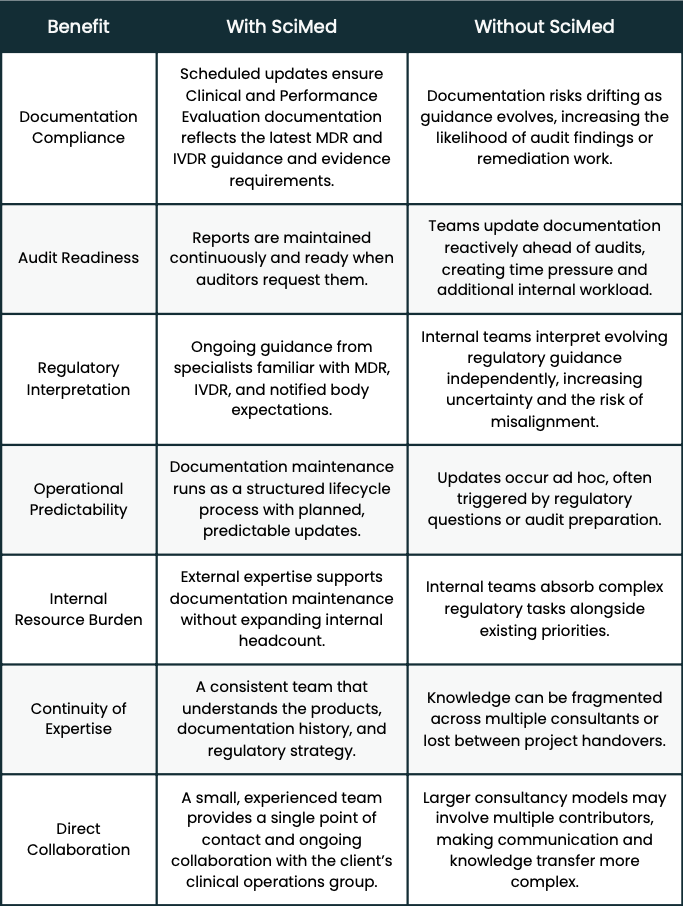
THE CHALLENGE: Compliance is a Continuous Commitment
Under both the MDR and IVDR frameworks, Clinical and Performance Evaluation documentation is a living obligation. Literature surveillance must be current. Regulatory guidance must be reflected. Real-world performance data must be incorporated. And throughout all of it, documentation must remain defensible; not just at the point of writing, but whenever a notified body or auditor decides to look.
For the clinical operations team at LifeScan, that created a very practical problem.
They had invested considerable effort in building compliant documentation across their portfolio. But sustaining that standard, continuously, across both MDR and IVDR frameworks, with multiple products, against a backdrop of evolving regulatory expectations - required a level of ongoing attention that was difficult to absorb internally without diverting resource from other critical work.
The risk of doing nothing was clear. Without a structured process, documentation could quietly drift out of alignment. Literature searches could age past the three-month threshold that regulators expect. Updates that should have been routine could stack up into something far more burdensome ahead of an audit.
What the team needed was a way to keep their evaluation documentation continuously MDR and IVDR-ready, predictably, without disruption, and without adding headcount.
THE SOLUTION: A Lifecycle Process, Not a Series of Projects
Following the initial implementation work, SciMed continued to support LifeScan through an ongoing literature monitoring and State-of-the-Art maintenance for Clinical and Performance Evaluation documentation..
The shift in mindset was important. Rather than treating documentation updates as occasional projects (triggered by audits, regulatory questions, or internal pressure) the collaboration was structured as a continuous lifecycle process. Reviews and revisions follow a defined schedule. Literature surveillance runs quarterly, ensuring searches never exceed a three-month window. Regulatory developments are tracked and incorporated as they emerge.
Both teams know precisely when each update is due, who is responsible for what, and what the output will look like. There is no scramble. There is no ambiguity.
Over time, that predictability has compounded. The SciMed team now operates as a genuine extension of LifeScan's clinical operations function, familiar with the portfolio, aligned on regulatory expectations, and able to move quickly when something changes.
The collaboration also delivered several operational advantages for LifeScan’s clinical operations team:
Need support maintaining MDR or IVDR Clinical and Performance Evaluation?
SciMed provides structured lifecycle support for Clinical and Performance Evaluation documentation, including Ongoing Literature Monitoring, State-of-the-Art updates, and audit preparation.
THE RESULTS: Audit Ready, Without the Anxiety
Today, LifeScan's Clinical and Performance Evaluation documentation processes are mature, current, and, critically, quiet.
Reports are updated on schedule. Literature searches stay within the required window. When a notified body or auditor asks a question, the clinical operations team can respond from a position of confidence rather than urgency.
The internal resource burden has also shifted. Rather than monitoring regulatory developments alone and interpreting what they mean for specific products, the LifeScan team can rely on SciMed's broader experience across multiple products and notified bodies. That collective knowledge shortens response times and reduces the risk of misinterpretation.
For a team with a wide portfolio and significant compliance responsibilities, that is a meaningful change to how they work day to day.
CONCLUSIONS: The Work Does Not End at Implementation
Regulatory compliance does not end once documentation is written. Under MDR and IVDR, keeping Clinical and Performance Evaluation documentation defensible requires a sustained commitment to evidence monitoring, State of the Art maintenance, and lifecycle management.
By building a structured post-market support model with LifeScan, SciMed has helped ensure that evaluation documentation remains current, aligned with regulatory expectations, and ready for scrutiny — without adding complexity to the day-to-day work of the clinical operations team.
It is, at its core, a straightforward objective: documentation that is always where it needs to be, whenever it needs to be there.
Keeping Clinical and Performance Evaluation documentation compliant shouldn’t consume your internal resources
If your team needs support maintaining Clinical or Performance Evaluation documentation under MDR or IVDR, SciMed can help.

![[LifeScan]](https://images.squarespace-cdn.com/content/v1/648ad45845bc567a651a5946/332dd1a0-f882-4272-a655-bd7ffde20dfa/client-logo-lifescan-colour.png)